Blogs

by Michael Cimaomo | Mar 1, 2019 | Northeast Underground
Potty Mouth SNAFU (Get Better Records) Release date: 3/1/19 They say time and distance heals all wounds. And on its second full-length album, the formerly Massachusetts-based trio Potty Mouth puts its own spin on this old adage while polishing up its alternative pop...

by Michael Cimaomo | Jan 10, 2019 | Blogs, Northeast Underground
To paraphrase Elton John, “This blog’s still standing!” Northeast Underground is celebrating its eighth anniversary today, and what better way to mark the occasion than by sharing the top eight most viewed posts from the blog’s entire run. Read below to see what...

by Michael Cimaomo | Sep 21, 2018 | Blogs, Northeast Underground
Various Artists BLAZE: Original Cast Recording (Light in the Attic Records) Release date: 9/21/18 Listening to the soundtrack of a film one has yet to see is a test for the ears. Free of images to pair with the music, the mind digests the sounds heard in as-is...

by Michael Cimaomo | Aug 13, 2018 | Northeast Underground
The Green Sisters Endless Blue (self-released) Release date: 6/30/18 Call it a family affair. Given these turbulent times, this debut release from four singing multi-instrumentalist sisters from Hubbardston – Rebecca, Betsy, Brianna and Melody Green – is a welcome...

by Michael Cimaomo | May 29, 2018 | Northeast Underground
According to a passage in his novel “Redburn,” no less an authority on seafaring life than Herman Melville states, “It is a great thing in a sailor to know how to sing well.” For, he explains, those at sea who are blessed with a talented voice may help raise the...

by Michael Cimaomo | Jan 10, 2018 | Northeast Underground
Seven is a tricky number. Lucky number seven, the seven deadly sins, “Seven Brides for Seven Brothers” – various attributes and symbols have been attached to this figure countless times. And no association with the number seven feels more applicable today than the...

by Michael Cimaomo | Sep 15, 2017 | Northeast Underground
Foo Fighters Concrete and Gold (Roswell Records/ RCA Records) Release date: 9/15/17 How does a stadium-filling rock band follow up its most ambitious album ever? In the case of the Foo Fighters, the answer is by achieving rebellion through an embrace of normalcy....

by Dave Eisenstadter | Apr 14, 2017 | Articles, Blogs, News, Newsletter
I had never read Noam Chomsky before or seen him speak, but I’d definitely heard about him over the years. Most recently when watching the movie “Captain Fantastic,” when the main characters — a super smart, back-to-the-land family — all celebrate “Chomsky...

by Michael Cimaomo | Apr 7, 2017 | Northeast Underground
LuxDeluxe Let’s Do Lunch (Spirithouse) Release date: 5/5/17 The latest release from these Northampton rockers is a pretty straightforward affair. In fact, the album’s opening track, “Intro,” even welcomes listeners and lists each band member’s name in succession. Ned...

by Michael Cimaomo | Mar 10, 2017 | Northeast Underground
Elliott Smith Either/ Or: Expanded Edition (Kill Rock Stars) Release date: 3/10/17 Either you are an Elliott Smith fan, or you are not. For some, Smith is the singer and songwriter who contributed a handful of heart-rending songs to the soundtrack of the film “Good...

by Chris Rohmann | Jan 9, 2017 | Articles, Arts, Blogs, Columns, Music, Newsletter, Stage, Stagestruck
Partway through last Friday’s performance at West Springfield’s Majestic Theater, something unscripted, overdue, and quite wonderful happened. The show was Peter Shaffer’s brilliant examination of genius and envy, Amadeus. The title refers to Mozart, but the...

by Michael Cimaomo | Jan 10, 2017 | Northeast Underground
Another year, another milestone. As of today, this blog is six years old. But what is there to make of this anniversary? Benchmarks of five years or 10 feel more momentous than just another notch added to the belt during the intervening period. And make no mistake...
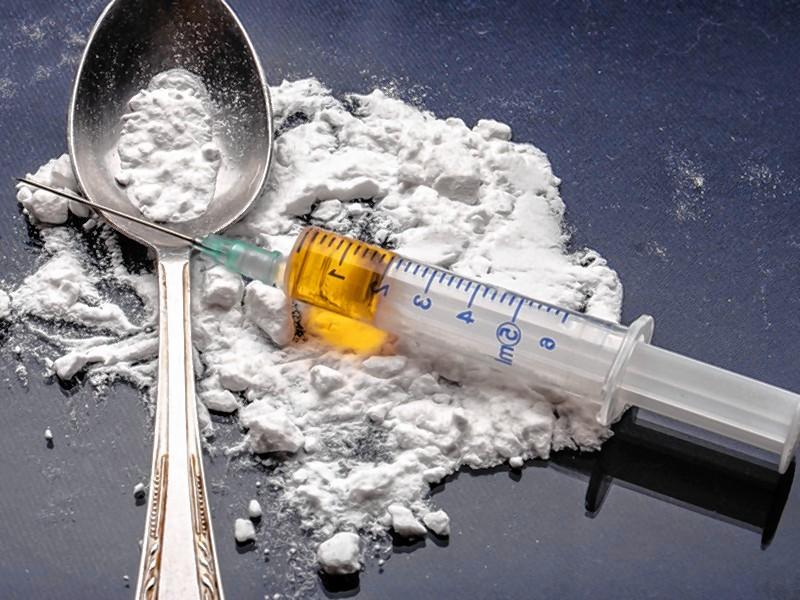
by Advocate Staff | Jan 6, 2017 | Articles, Blogs, News
Each week, the Advocate staff assembles for an AdvoChat, where we bat around thoughts on a subject in the news. The chat is lightly edited. dave.eisen (Dave Eisenstadter, web editor): Kristin, you’re working on a piece on addiction (EDITOR’S NOTE: here it...

by Michael Cimaomo | Aug 5, 2016 | Articles, Featured, Northeast Underground
Dinosaur Jr. Give A Glimpse Of What Yer Not (Jagjaguwar) Release date: 8/5/16 Break out the ear plugs. Western Massachusetts’ own alt-rock power trio, Dinosaur Jr., is back. New album Give A Glimpse Of What Yer Not marks the fourth release by the band since the...

by Michael Cimaomo | Aug 12, 2016 | Northeast Underground
Indian Oven Tenderness (Self-release/ DIY) Release date: 8/12/16 In a 2013 interview, the members of the Western Massachusetts indie rock band Indian Oven – Sam Carpenter (vocals, guitar, bass) and Griffin Bazzeghin (drums, percussion, harmonica, backup vocals) –...

by Michael Cimaomo | Jun 4, 2016 | Newsletter, Northeast Underground
And The Kids Friends Share Lovers (Signature Sounds) Release date: 6/3/16 In the midst of graduation season in Western Mass, it’s easy to forget that graduation itself is about more than caps and gowns. Students, who four years ago were mere freshmen in high school or...

by Michael Cimaomo | May 10, 2016 | Articles, Arts, Blogs, Music, Northeast Underground
House shows have always been an important part of the DIY music scene. Whether it’s some garage rockers providing the soundtrack to a backyard party, a punk rock band playing a concert in a basement or even some indie folk musicians holding court in a living room, the...

by Michael Cimaomo | Jan 10, 2016 | Northeast Underground
It’s been written before that five years is a long time. And after five years of Northeast Underground blog posts (over 200 articles and counting!), few could imagine just how much has been covered, discussed and critiqued on this little slice of the Internet. So on...

by Hunter Styles | Dec 14, 2015 | Articles, News, The Uncanny Valley
Like many local drivers, I take Interstate 91 up and down the Valley several nights a week. So I have had plenty of opportunity to wonder: what in the intergalactic hell is going on near exit 24? Drive south for a mile and a half past the Whately Diner, look to the...

by Michael Cimaomo | Dec 15, 2015 | Articles, Arts, Music, Northeast Underground
This isn’t your parents’ best of list. To paraphrase Jimmy Fallon, “You know those awards they give out in high school yearbooks? Awards like Most Likely to Succeed, Class Clown, stuff like that?” Well, Northeast Underground is handing out its own superlatives this...

by Michael Cimaomo | Nov 20, 2015 | Northeast Underground
Blue Jean Committee Catalina Breeze (Drag City) Release date: 11/20/15 In November of 2011, the national entertainment spotlight turned its eyes towards Northampton, Mass. The reason for the attention was a sketch on Saturday Night Live. Featuring actor Jason Segel...

by Amanda Drane | Oct 26, 2015 | Blogs, News, The Uncanny Valley
After my colleague Hunter Styles dispelled the myth that the characters in the Scooby Doo TV series were based on the academic personalities of the Valley’s Five Colleges, my attention should have immediately turned to another legend passed on to me when I was a wee...

by Hunter Styles | Oct 7, 2015 | Articles, Blogs, Featured, The Uncanny Valley, Uncategorized
by Hunter Styles Staff writer Amanda Drane has worked in the local food industry for 10 years, and she’s shared with us some truly great stories of her time in the kitchen, including a couple about the haunted room at Spoleto, an Italian restaurant in Northampton. One...

by Kristin Palpini | Sep 8, 2015 | Articles, Blogs, Columns, Featured, News, The Uncanny Valley
Why is it that a cheeseburger costs $1.22 at Burger King, while a small order of fries costs $1.70? Isn’t beef supposed to be more expensive than potatoes? Thanks for writing in, anonymous reader, with your puzzling — and discomforting — question. To figure out why...
by Michael Cimaomo | Aug 3, 2015 | Northeast Underground
Jesse French is not your average gardener. Though by day, the Hampshire College graduate works for the Trustees of Reservations in Holyoke, Mass. doing landscaping and maintenance, by night the musician inside French comes out to play. From jobs doing live sound at...

by Amanda Drane | Jul 28, 2015 | Articles, Blogs, Featured, News, The Uncanny Valley
Buses are big and so are the advertisements they often host — that’s no surprise. What is jolting, however, is when the typical music or beverage ad is replaced with a giant placard offering a $50,000 reward for information regarding an unsolved murder from 2013....

by Amanda Drane | Jul 14, 2015 | Blogs, News, The Uncanny Valley
My curiosity is piqued by a recent Facebook post by fellow Advocate writer Yana Tallon-Hicks in which she documented a creepy, abandoned house complete with a puzzling sign that read, “This is my happy place.” So, I head past the rolling greens of the Amherst Golf...

by Kristin Palpini | Jun 30, 2015 | Articles, Blogs, Featured, News, The Uncanny Valley
You’ve probably seen the signs — “Fast $$ for Houses,” “We Buy Ugly Homes” — tacked onto telephone poles or scrawled onto yard signs by the side of a main road. The advertising doesn’t inspire confidence. The hand-scrawled, occasionally misspelled signs scream scam....
by Michael Cimaomo | Jun 19, 2015 | Northeast Underground
The music festival, once a haven for the young, rebellious and sometimes nude, has become a family affair. Forget Coachella, forget Bonnaroo, and forget Lollapalooza. This summer Western Massachusetts plays host to three different music festivals, all billed as prime...

by James Heflin | Jun 9, 2015 | Articles, Arts, Blogs, Columns, Featured, Leisure, Music, News, Nightcrawler
WEB TEXT FOR ARTS PAGE 1

by Kristin Palpini | Jun 10, 2015 | Articles, Blogs, Featured, News, The Uncanny Valley
For more than a decade, the island at the corner of Hampton Street and Route 5 in Holyoke has borne the word “Quota” — spelled out in flowers in the warm weather and black plastic edging in the cold. On my ride home from work down Route 5 south, I often wondered...

by James Heflin | May 27, 2015 | Articles, Blogs, Featured, News, The Uncanny Valley
Spring-Heeled Jack, in addition to being the best-named apparition since the Mad Gasser of Mattoon, was a frequent haunter of Victorian London and, eventually, other parts of Great Britain. He was known and feared for his habits of sudden attack via tearing with metal...

by Kristin Palpini | May 27, 2015 | Arts, Blogs, News, The Uncanny Valley
Behind the scenes at the Wistariahurst Museum in Holyoke in the 1990s, Stephen Petegorsky made a delightful discovery: “hundreds of taxidermed animal specimens — most of them really ratty — stuffed in a storage closet.” He borrowed some stuffed animals for photo...

by Kristin Palpini | May 27, 2015 | Articles, Arts, Blogs, Featured, News, Wellness
On occasion, to keep up my health, I take a walk. Nearby my home in West Springfield is a mile-long stretch of sidewalk with no hills, few cars, and even fewer people outside. The homes are densely packed, and I wonder about the people who live inside. Over time, I’ve...

by Kristin Palpini | May 20, 2015 | Blogs, News, The Uncanny Valley
Reporter Amanda Drane recently turned me on to this weird phenomenon that’s been recorded around the world. Out of nowhere, in the day or night, in the city or in the woods people have recorded the incredibly loud sound I can only describe as a giant sky whale...

by James Heflin | May 12, 2015 | Articles, Arts, Between the Lines, Blogs, Columns, Featured, Leisure, Music, News, Nightcrawler
Back in the final year of the 1900s, I stood, guitar in hand, on the steps of Northampton’s old courthouse at the main intersection. The occasion was the (then new) Valley Advocate Grand Band Slam. My bandmates and I had won top honors in the...

by James Heflin | May 12, 2015 | Arts, Blogs, Music, News
Usually, it’s a happy occasion that puts a band in the paper. This week, it’s tragic news. As you may have heard, The Alchemystics recently lost two of their circle. Drummer Demse Zullo and his longtime friend Brian White were killed when the van they were travelling...

by Amanda Drane | May 12, 2015 | Articles, Blogs, Featured, News, The Uncanny Valley
On the evening of March 17, a driver in South Hadley reported seeing a strange triangular craft in the sky with lights on each of its three visible points. After following the craft for a couple of miles, the driver pulled into the Village Commons parking lot and...

by Amanda
Drane | Apr 28, 2015 | Articles, Featured, Free Sport, News
Summer is just around the bend and the Connecticut River — the region’s longest body of running water — remains largely unswimmable due to high levels of fecal bacteria in the water. The river has come a long way since its days nicknamed “America’s most beautiful...

by Hunter Styles | Apr 28, 2015 | Articles, Columns, Featured, News, The Uncanny Valley
“Curiouser and curiouser!” exclaimed Alice as she took her first steps into Wonderland. If she were making a trek through the Valley instead, we think she would say the same thing. Our little corner of the globe is chock full of odd people, secret places, and...

by Hunter Styles | Apr 7, 2015 | Articles, Featured, Free Sport, News, Wellness
Marla Brodsky’s boots crunch on the snowy path. In the dog yard behind her house, 18 pairs of ears perk up at the sound. As she approaches, her four litters of Alaskan Husky sled dogs start to stir. Some dogs lift their heads and howl. Some strain at their leashes....

by Amanda Drane
and Hunter Styles | Apr 1, 2015 | Articles, Arts, Blogs, Featured, Free Sport, Leisure, News, Wellness
Canalside Rail Trail, Turners Falls A handful of beautiful sights crop up along the four miles of this short, scenic bike path, which runs from Deerfield up into Montague along old railroad beds. But the short northern stretch where wooded areas gives way to the...

by Amanda Drane | Apr 1, 2015 | Articles, Blogs, Featured, Free Sport, Leisure, News, Wellness
His players say Winston Lee’s life revolved around baseball and softball. When he died in 2011, the players in his Spanish American Softball League were devastated. Hundreds of the inner-city players not only missed the man, but the community he’d built around him....
by Michael Cimaomo | Mar 28, 2015 | Northeast Underground
Lady Lamb After (Mom + Pop) Release date: 3/3/15 It’s part of pop culture lore that Quentin Tarantino got his start working in a video store. Turns out, that watching innumerable films and critiquing customer choices was all the training he needed to become a famous...

by Jack Brown | Mar 24, 2015 | Arts, Blogs, Cinemadope, Columns, Film
Everyone with a go-to Chinese take-out joint is sure of one thing: theirs is the best. It’s a partisan game on a par with Left vs. Right or The Beatles vs. The Stones — I once witnessed, on a Brooklyn street corner, a heated argument about the neighborhood’s best...

by Gary Carra | Mar 24, 2015 | Arts, Blogs, Columns, Leisure, Music, Nightcrawler
She’s seen fire. She’s seen (freezing) rain. But Kristy Librera Chapman remains undaunted in her quest to aid her family and her community. A cousin to one of the families affected by a tragic Christmas Eve fire in Southwick, Chapman says that she knew she needed to...
by Sarah Werthan Buttenwieser | Mar 20, 2015 | Blogs, Sitting On The Baby, Uncategorized
One thing that I found especially shocking when I became a first-time mama to a boy was the assumption that boys are genetically programmed to like trucks and balls and big creatures like dinosaurs. Some boys adore all of the above. Some don’t. The fact that my...
by Sarah Werthan Buttenwieser | Mar 4, 2015 | Standing In The Shadows
Ellen Meeropol dreamt up a horror. She took a charged anniversary, September 11th, a remote island in Maine, and an assortment of characters whose agendas are at odds (at the most polite best), but she doesn’t go for “at best” in her second novel, On Hurricane Island,...
by Advocate Staff | Feb 26, 2015 | The Public Humanist
Cutting to the chase: The Public Humanist’s current and archived posts can now be found here: http://masshumanities.org/the-public-humanist/ And now to back up a little. Many of you will have gleaned that The Public Humanist is the one blog on the Valley Advocate site...
by Michael Cimaomo | Feb 24, 2015 | Northeast Underground
Iron & Wine Archive Series Volume No. 1 (Black Cricket Recording Co.) Release date: 2/24/15 Dubbed as “the first in a series of releases pulled from tapes found in the back corners of closets and dusty shoe boxes – long neglected, but never forgotten,” the newest...

by Chris Rohmann | Feb 5, 2015 | Blogs, Columns, Stagestruck
They just don’t make musicals like they used to. Except when they do. Nice Work If You Can Get It, playing at the Bushnell in Hartford through February 8th, is a 1920s musical that premiered on Broadway in 2012. It was whipped up by Joe DiPietro from the skeleton of a...

by Caleb Rounds | Jan 28, 2015 | Blogs, Leisure, Talk Dirt to Me
Most of us in this country generate a lot of trash. I spend a lot of time feeling guilty about it, but it turns out that doesn’t actually decrease my trash production. With a tiny amount of effort I can recycle all the paper and plenty of the glass and plastic. Even...
by Michael Cimaomo | Jan 28, 2015 | Northeast Underground
In 1976, the Ramones charged out of New York City proclaiming, “I Don’t Wanna Go Down to the Basement.” “There’s something down there,” sang lead singer Joey, with a delivery that illustrated his reluctance and dread. Now almost 40 years later, Greenfield band...

by Caleb Rounds | Jan 21, 2015 | Blogs, Leisure, Talk Dirt to Me
I, like most everyone who can read this, am a proud endotherm. I do a pretty good job of maintaining a constant internal temperature. It’s something I’m pretty good at, so why shouldn’t I be proud? All birds and mammals are endotherms. There are even some large fish...
by Caleb Rounds | Jan 22, 2015 | Blogs, Talk Dirt to Me
Most of us in this country generate a lot of trash. I spend a lot of time feeling guilty about it, but it turns out that doesn’t actually decrease my trash production. There’s very little incentive for cutting down other than ideology. With a tiny amount of effort I...

by Caleb Rounds | Jan 15, 2015 | Blogs, News, Talk Dirt to Me
We’re in the midst of the good New England weather that keeps the weak away. It looks desolate and lifeless out there, but it most surely isn’t. With temperatures staying below freezing the little snow we have is sticking around and treating us to the signs of the...
by Caleb Rounds | Jan 13, 2015 | Blogs, Talk Dirt to Me
We’re in the midst of the good New England weather that keeps the weak away. It looks desolate and lifeless out there, but it most surely isn’t. With temperatures staying below freezing the little snow we have is sticking around and treating us to the signs of the...
by Michael Cimaomo | Jan 10, 2015 | Northeast Underground
Four years – the space between Olympic Games, the duration of the average college career, or the amount of time some bands take to record and release a new album. If you, dear reader, told me four years ago that I would still be writing this blog in 2015, I would’ve...
by Sarah Werthan Buttenwieser | Jan 4, 2015 | Standing In The Shadows
The stirrings of January and it’s apparently the year of the non-resolution, by which I mean people seem to be in exploration of the notion that we are enough as we are or less and more are ways to beat up on ourselves (read more about this on Abigail Rose...

by Caleb Rounds | Jan 7, 2015 | Blogs, News, Talk Dirt to Me
This winter we haven’t gotten enough snow. “Enough” is what allows for cross-country skiing. Even in New Hampshire, where I spent part of the holidays, the snow cover was patchy or absent. We passed a wet hour tubing at a ski resort. The wet snow and underinflated...



















































